SP1 - SYMPATH
Clinical and epidemiological cohorts, data integration, central database, and project support
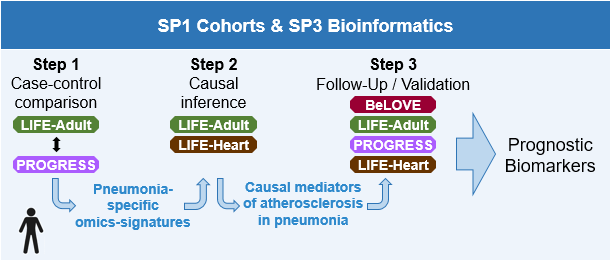
In subproject 1 of the SYMPATH consortium, data from selected volunteers and patients from large studies will be used to comprehensively characterize the course of atherosclerosis after pneumonia. In patients enrolled in the PROGRESS study for pneumonia, data will be collected on subsequent atherosclerotic cardiovascular events such as myocardial infarction or long-term stroke. Accordingly, in patients with cardiovascular disease, information on past pneumonia is collected in the LIFE-Heart study. In subjects of the LIFE-Adult population study, clinical data on both diseases are supplemented. In addition to the clinical and phenotypic findings on the diseases, the participants in these studies also have comprehensive biomaterials at their disposal, which are further analyzed within the framework of SYMPATH using multiomics methods and integrate the data obtained into further characterization. These analyses include DNA sequencing, RNA analysis and measurements of proteins, cytokines and metabolites. The molecular data will be processed together with the clinical data on pneumonia and cardiovascular diseases and risk factors of patients and volunteers for the identification of new mediators and mechanisms of pneumonia-aggravated atherosclerosis and made available by means of a database for the work in subproject 3. New data from the Berlin long-term observational study for vascular events - BeLOVE - will be made available to review the findings of subproject 3. In this high-risk cohort the prognostic relevance of the molecular signatures will be validated with the aim of an improved risk stratification of pneumonia-associated cardiovascular events.