TP4 - SysMedSUDs
In silico prediction of dynamical alterations in multiscale neurochemical connectomes in SUDs
Despite diverse molecular mechanisms of action, drugs of abuse affect distinct yet partially overlapping neurocircuitries. We have demonstrated that craving for psychoactive substances relates to the activation of specific brain-wide networks, yet the underlying neurobiology remains elusive. In this subproject, we apply mathematical modelling and simulations to investigate how acute and chronic exposure to different drugs (alcohol, nicotine, THC, heroin, and cocaine) changes cellular activity, particularly in terms of short-term and adaptive alterations in neurotransmitter concentrations. Thereby, our mathematical model comprised by a set of coupled nonlinear delay-differential equations makes use of two massive databases. By screening 338,982 publications, we have systematically collected data of 147,138 rats, which has been integrated into two open-access databases; www.chemnetdb.org and www.syphad.org. By superimposing the connections, neu-rochemistry, and cell types in a terminologically and mathematically consistent manner on ana-tomical data, ChemNetDB provides a unique comprehensive framework for the effective study of the normal and diseased brain. Thus, it will serve as the topological foundation of our model. The Syphad database contains detailed neurochemical fingerprints of psychiatric drugs and will be used as the basis for machine learning algorithms for optimal identification of model parameters. For different patterns of drug intake and different concentrations, our patented in silico procedure predicts for each brain region, the temporal trajectories of concentrations of various neurotransmitter, particularly amino acids, monoamines and neuropeptides. This neurochemical decoding of drug-induced changes in rat brain improves our understanding of the distinct, overlapping and time-dependent mechanisms of action of drugs of abuse at systems level, and paves the way for development of optimal treatment strategies for relapse prevention.
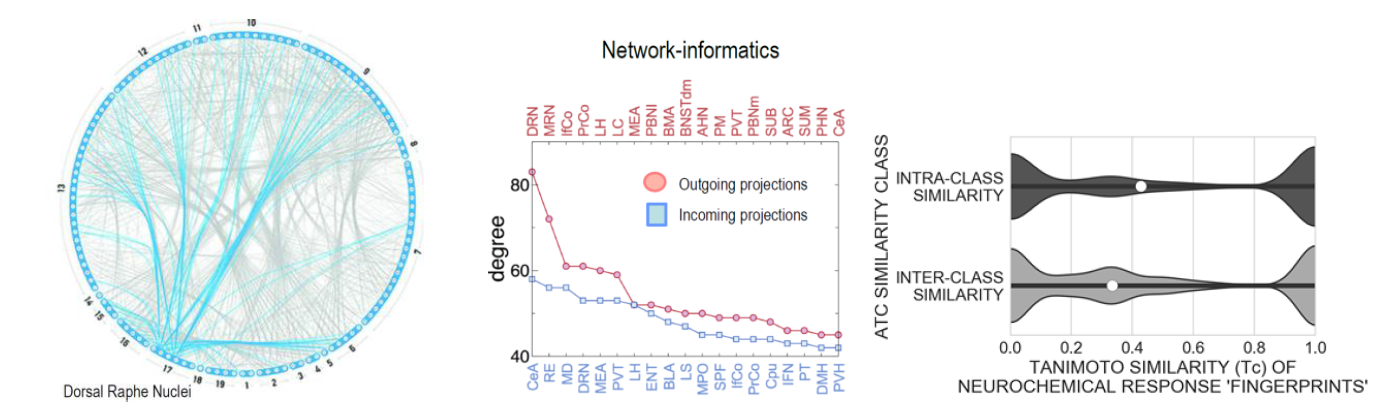

The neurochemical (neurotransmitter-based) connectome of the rat brain and its network characteristics and neurochemical fingerprints of psychiatric medication (described in detail in our recent studies by Noori et al., 2017; 2018). Left part: Schematic representation of the neurochemical connectome by a connectogram with 125-nodes (brain regions) partitioned in 19 large-scale brain regions. Depicted are only serotoninergic connections that derive mainly from node raphe nuclei the source of serotoninergic fibers. Middle part shows graph properties with in- and out-degree ranking. For example the dorsal raphe nuclei (DRN) has > than 80 outgoing projections and the central amygdala (CeA) has most of the incoming projections within the rat brain. Right part shows intra- and inter-ATC code (ATC refers to the Anatomical Therapeutic Chemical Classification System) similarity of neurochemical response patterns across brain regions (median similarities are identified by the white marker). Neurochemical fingerprints will be also calculated for all 5 drugs of abuse after long-term self-administration in the reinstatement paradigm allowing us to make quantitative conclusions about their similarities.



