SP 1
Sample management, monitoring of viral infection and virus-specific immunity
During the last decades, acute rejection and allograft survival rates have been significantly improved by the introduction of new potent immunosuppressants. The price in many patients is, however, over-immunosuppression resulting in side effects (nephrotoxicity, opportunistic infections such as EBV, CMV, BKV, and other; malignancies, or metabolic disorders) and high costs. Previously used protocols aimed at minimization of immunosuppression may avoid side-effects but are insufficient in preventing acute rejection in some patients. In our previous studies, we showed that reactivation of latent viruses such as CMV, EBV, and BKV can lead to a graft dysfunction, and should be clearly distinguished from acute rejection for the initiation of appropriate therapies. In some cases, viral (BKV) reactivations can mimic acute rejection and differential diagnosis between infection and acute rejection is not possible by standard methods. Very recently we established a high-throughput method of T cell analysis by next-generation sequencing (NGS) of T cell receptor (TCR). Based on this new methodology we tracked BKV-specific T cells in blood, urine and transplant tissue and provided ultimate diagnostic help for therapeutic decision.
Within e:Kid we also handle, store and provide clinical samples to consortium partners. A well-characterized patient cohort of 500 renal transplant patients will serve as a basis for the collaborative project. Furthermore, we will perform viral load analysis of EBV, CMV and BKV as well virus-specific T cell monitoring by multi-parameter flow cytometry and TCR-NGS that can be helpful predicting clinical course of viral infection. The data will be transferred to bioinformatics partners for mathematical analyses. Established biomarkers and models will provide a basis for initiation of a prospective risk-adjusted clinical trial on personalized immunosuppressive regimen (second funding period).
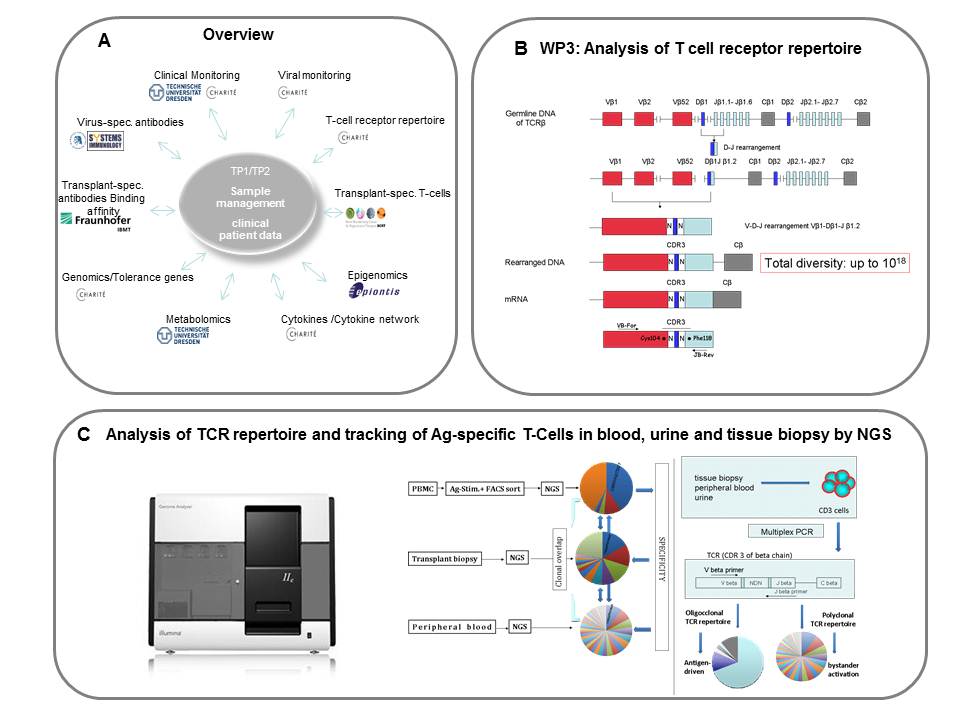
A) Overview of the activities within the subproject: providing consortial clinical sample management, viral (EBV, CMV, BKV) load and antiviral-specific immunity analyses;
B) Rearrangement of TCRβ locus within CDR3 fragment determines clonality of T-cells;
C) Analysis of TCR repertoire and tracking of Ag-specific T-Cells in blood, urine and tissue biopsy by NGS.
Keywords: Systems medicine, kidney transplantation, immunosuppressive



