SYS-GLIO
Systems‐based predictors for the biological and clinical behavior of gliomas
Diffusely infiltrating astrocytic gliomas are the most common intrinsic brain tumors and cover a spectrum of malignancy grades, ranging from diffuse astrocytoma (WHO grade II) over anaplastic astrocytoma (WHO grade III) to glioblastoma (WHO grade IV). Clinically, two types of glioblastomas are distinguished, i.e. primary glioblastomas that arise de novo with a short clinical history, and secondary glioblastomas that develop by progression from a preexisting lower grade glioma. Despite sharing similar histological features, primary and recurrent glioblastomas carry distinct genomic, epigenetic and transcriptional aberrations for review see. Glioblastomas carry a universally dismal prognosis in children and adults with median survival times still below 12 months from diagnosis. In this project we want to analyze the genetic characteristics of malignant brain tumors of different WHO grade greater detail. With the results of these investigations we want to develop new strategies to stop the malignant progression of low-grade gliomas and prevent drug resistance in high grade lesions.
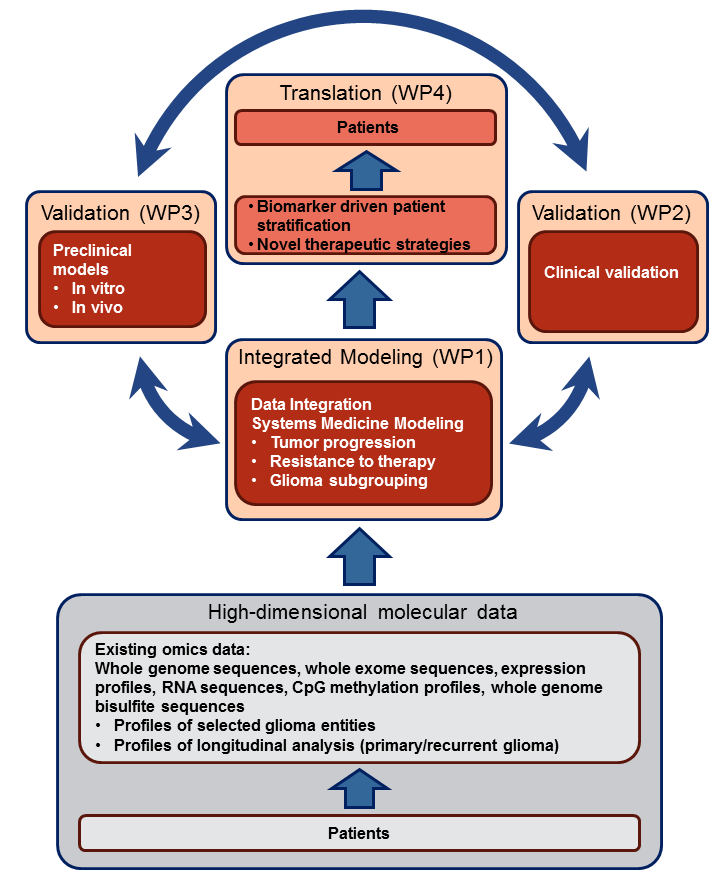
Subprojects in SYS-GLIO:
SP 1A Integrative analysis of genome-wide data sets
SP 1B Mathematical modeling of glioma growth
SP 2A Assessment of crucial pathways in validation cohort
SP 2B Assessment of crucial pathways in validation cohort
SP 3A In vivo validation in glioma mouse models
SP 3B Experimental modeling of glioma progression and therapy resistance
SP 3C Metabolic adaptations in glioma progression and therapy resistance



