SP1 - GUIDE-IBD
Central coordination of the clinical observation trial
In focus of the collaborative project GUIDE-IBD stands subproject SP1, which organizes the clinical observational study. The aim is to investigate, whether the additional molecular knowledge (novel molecular markersets) improve medical therapy decisions for patients with IBD receiving anti-TNF-biologics (dosage of medication, disease control). This task is controlled by three partners from Kiel (SP1a), Hamburg (SP1b) and Hanover (SP1c) while Kiel acts as the leading partner.
In total, 130 patients with IBD, such as Crohn's disease or ulcerative colitis, will be recruited, receiving anti-TNF therapy for the first time (infliximab or a similar drug (biosimilars)). The indication is decided by a local therapy conference, which takes place without any influence from the research program and therefore outside the study protocol. Only after therapy decision the patients will be asked for consent to participate in this study. The patient will then be randomly assigned to one arm of the study using a defined stratification and randomization algorithm. 65 patients are assigned to the so-called "molecular medicine" arm, in which previously validated marker panels on various data levels (gene expression, microbial markers, serum markers, innovative pharmacokinetics) will be analysed and provided to the treating physicians. These additional patient data are available to the doctor for his further therapy decisions. The other 65 patients receive the standard therapy, the so-called "best standard care", which is controlled according to current recommendations but without additional molecular data. In this group, we plan to access biomaterials as well, which will be analysed in the other subprojects (SP2 and SP4), but without further communication of the results within the clinical study. Subproject SP3 uses patient reported outcomes to determine possible new endpoints for clinical trials (continuous tracking of life quality profiles and physical activity using “wearables” and mobile apps). The materials for sampling are provided centrally by leading partner Kiel (SP1a). The organization of the samples is carried out in Kiel (SP1a), Hamburg (SP1b) and Hanover (SP1c) by technical / medical assistance.
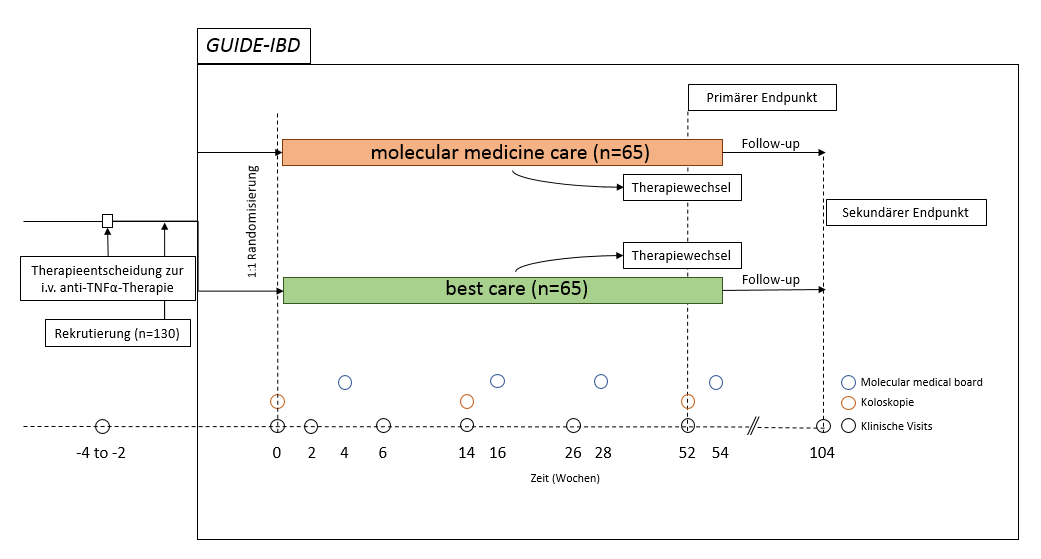
Scheme of patient recruiting and biomaterial sampling for the moleculare reporting. At week 4, 16, 28 and 54 post induction, the molecular data will be included into the therapy decision within the Molecular Medicine Board.



