SP 6
Clinical trial
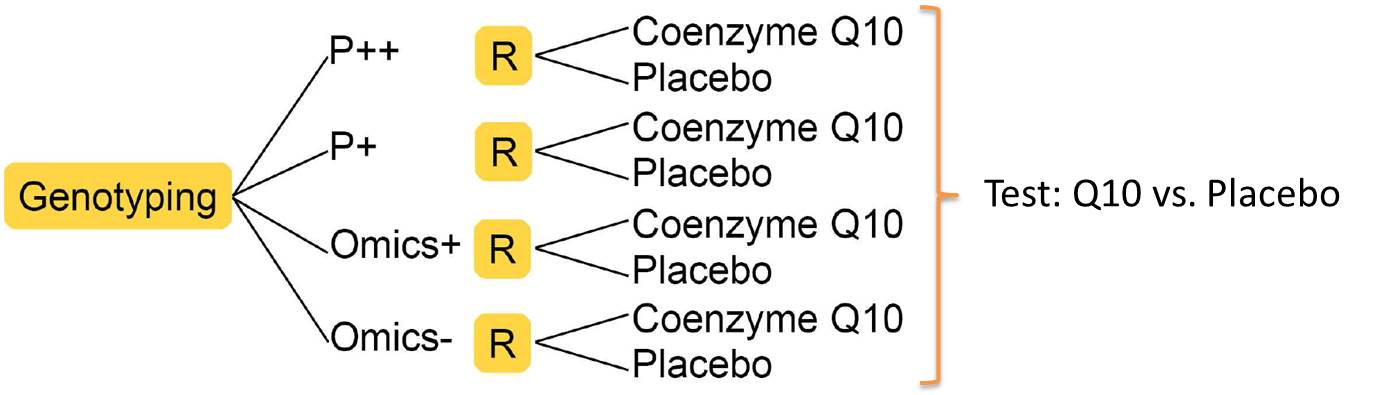
The aim of this subproject is to investigate the influence of coenzyme Q10 for the treatment of patients with idiopathic Parkinson disease (PD) depending on the patient’s mitochondrial dysfunction. Previous studies have reported a positive influence of Q10 to the mitochondrial function. Thus, it is supposed that the effect of Q10 treatment will increase with increasing degree of mitochondrial dysfunction of the patient. Therefore, the role of Q10 as a mitochondrial enhancer will be investigated in a bicentric, blinded, controlled and randomized clinical trial. PD patients will be divided into four subgroups depending on the degree of their expected mitochondrial dysfunction. PD patients with mutations in Parkin or PINK1 genes are expected to have highest mitochondrial dysfunctions. Further PD patients with mitochondrial dysfunction will be identified by an innovative “omics” approach. Study participants will be investigated depending on their genetic profile and divided into one of the following subgroups:
- P++: two mutations (homozygote or compound heterozygote) in Parkin or PINK1
- P+: one mutation in Parkin or PINK1
- Omics+: likely mitochondrial dysfunction, no mutation/s
- Omics-: unlikely mitochondrial dysfunction, no mutation/s
Study participants will be randomized within these subgroups to either Q10 or to the control group (see Figure 1).
The primary endpoint of this study is the difference of change of motor functions assessed by part III of the „Unified Parkinson Rating Scale“(UPDRS III) between Q10 and placebo group after 6 months. Secondary endpoints are e.g. MRSI measurements, 10-Meter-Walk-Test, quality of life (PDQ39), depression (BDI) and fatigue (FSS).
Optionally, the main trial will be followed by an open observational trial exploring the effect of vitamin K2 on the motor functions of PD patients.



