SP 3
TIL repertoire in melanoma and pancreatic ductal adenocarcinoma
Tumor-infiltrating lymphocyte (TIL) therapy has been pioneered and optimized in patients with advanced melanoma, where it can achieve an overall clinical response rate of >50%. Retrospective analyses of the infused T-cells have revealed that in vivo anti-tumor activity was associated with a higher number of infused cells, short time in culture, high frequency of CD8+ T-cells and possibly certain subsets of memory T-cells. While many studies provide the characteristics of the infused T-cells, little effort has been put into characterizing the TILs ex vivo and during expansion to understand how the T-cell infusion product compares to the naturally recruited repertoire. We propose to use T-cell receptor (TCR) repertoire analysis as a biomarker of TIL quality and clinical response.
It is conceivable that in the ≈50% of patients not responding to TIL therapy, TILs are of insufficient potency, or tumor-reactive clones are not maintained during culture, e.g. due to their exhaustion after in vivo antigen encounter. These patient subgroups could benefit from in vivo manipulations that improve the TIL available for expansion or optimized culture conditions preferentially expanding tumor-reactive cells.
We have recently found that TIL can also be isolated with high efficiency from biopsies of patients with pancreatic ductal adenocarcinoma (PDA) (see Fig.), a cancer type for which novel treatment approaches are urgently needed. We therefore aim to develop TIL therapy as a treatment strategy in PDA.
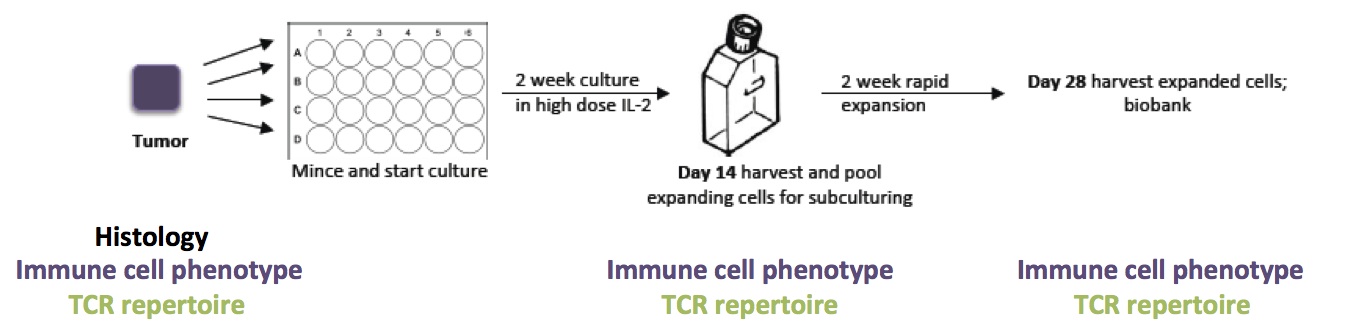

T cells from tumor biopsies are grown in presence of the growth factor interleukin 2. After two weeks of culture all expanding cells are pooled and expanded to large numbers during the following two weeks. T-cell surface and activation markers as well as the T-cell-receptor repertoire are measured during the culture and directly in the tumor and provide information on the "quality" of the culture.
Subproject 3 builds on close interactions with the other TIL-REP partners and will pursue the following aims:
- In-depth characterization of ex vivo and expanded PDA TILs by phenotype and TCR repertoire analysis in comparison to melanoma TIL (see Fig.)
- Exploitation of TCR profiling as biomarker for cancer immunotherapy trials
- Exploitation of TCR profiling to study TIL repertoire heterogeneity in tumor biopsies
- Biomarker-guided optimization of TIL culture strategies
Ultimately, we want to determine whether PDA-TIL lend themselves to clinical application and develop such therapies in collaboration with the European pancreas center at Heidelberg University hospital.



