e:Kid
Systems medicine approach to personalized immunosuppressive treatment at early stage after Kidney Transplantation
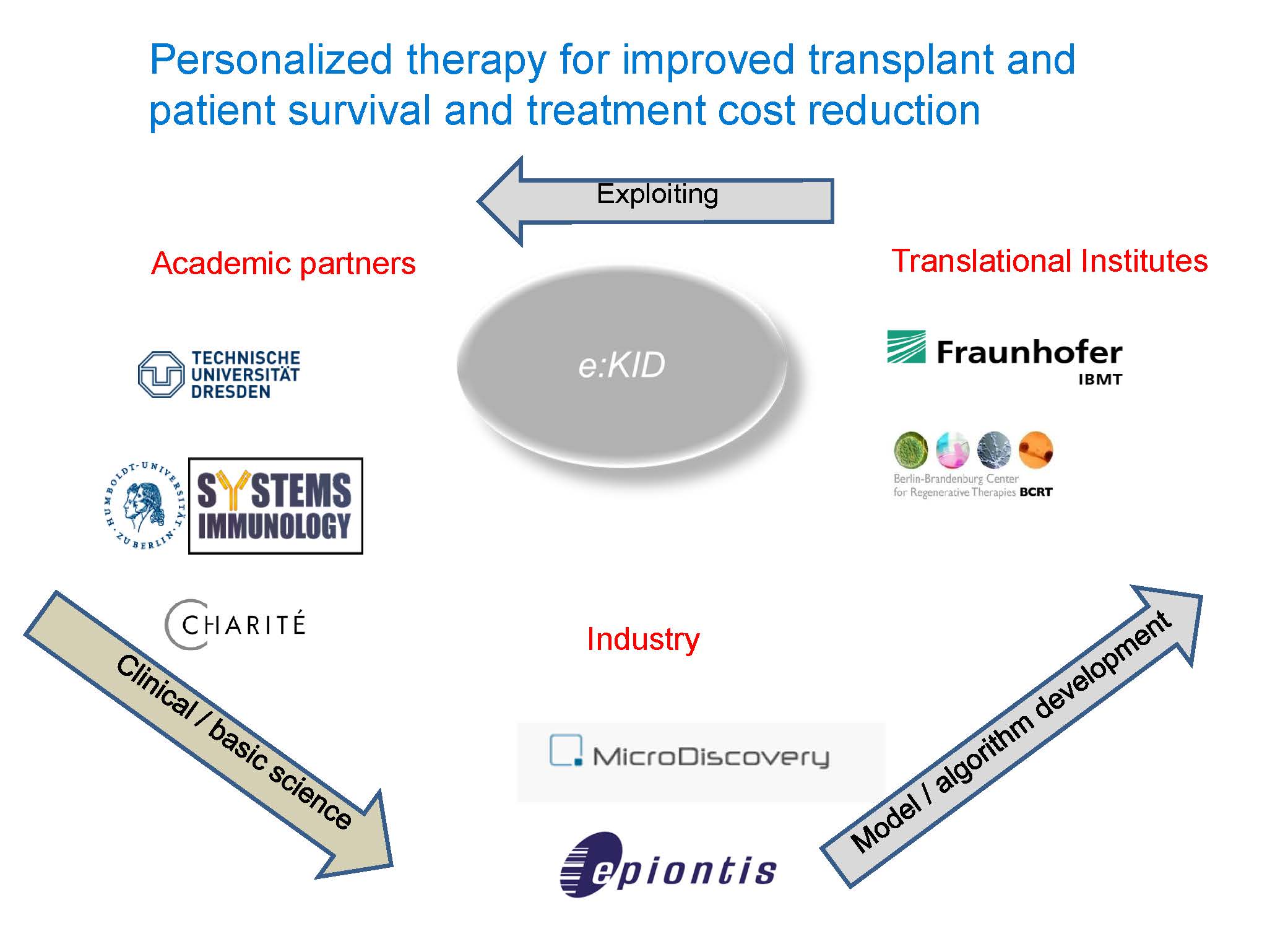
e:Kid is a collaborative project involving clinical departments, research institutes, biometrics departments, translational, and industrial partners. Several clinical and fundamental science based departments of Charité and the Technical University of Dresden, Department of Biology at the Humboldt University, and two industrial partners are involved in this project.
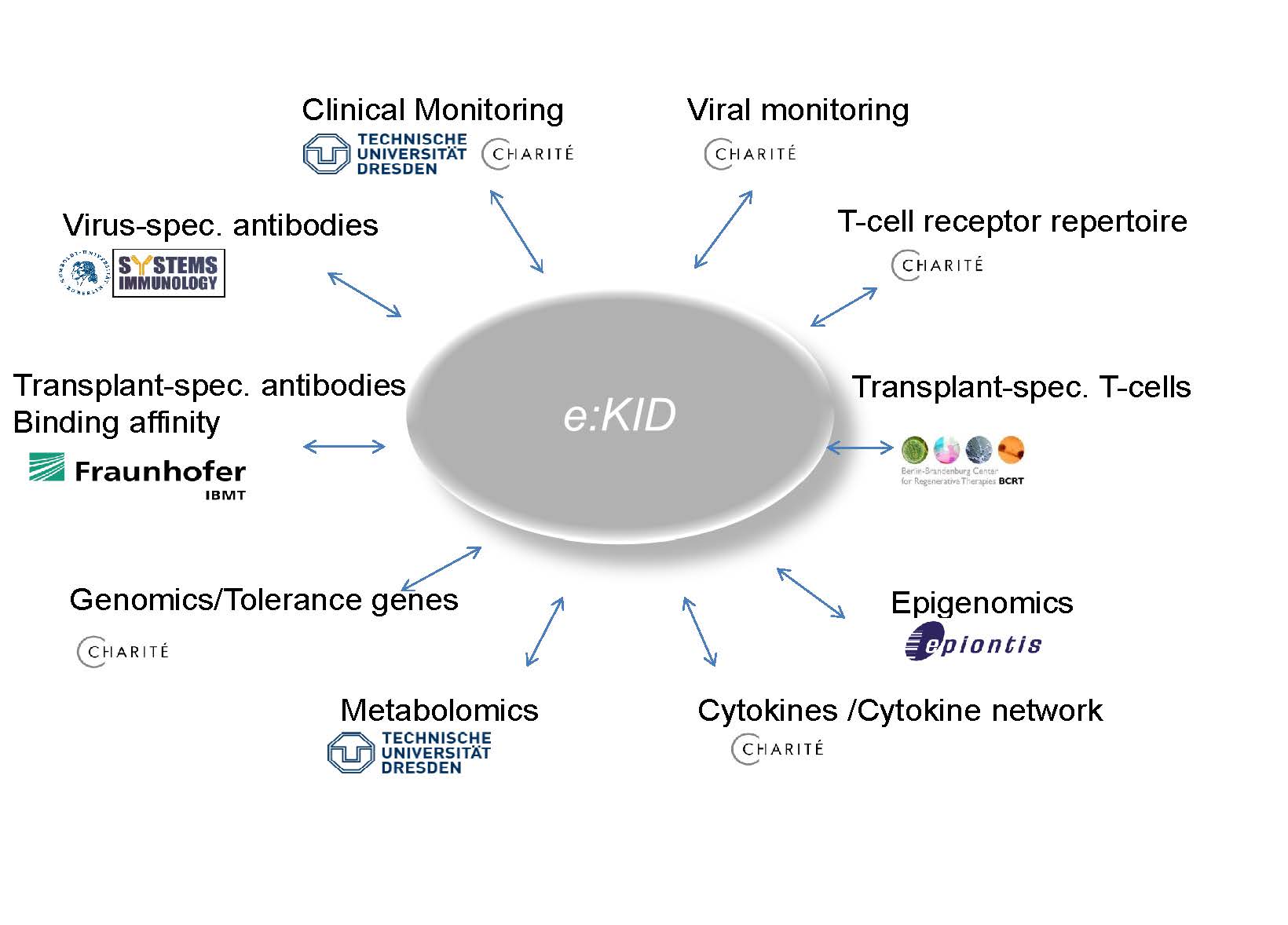
The aim of e:Kid is creating and validating a systems medicine based model allowing personalized therapy at early stage after kidney transplantation. Based on the expertise of the consortium members, a wide-range of different parameters on genetic, posttranslational (epigenetic, protein) and cellular level is examined in a large clinically well-characterized cohort of patients and used to analyze immunological pathways and to develop predictive mathematical models. These models enable an individual's risk for rejection after a kidney transplant or to predict transplant-related side effects.
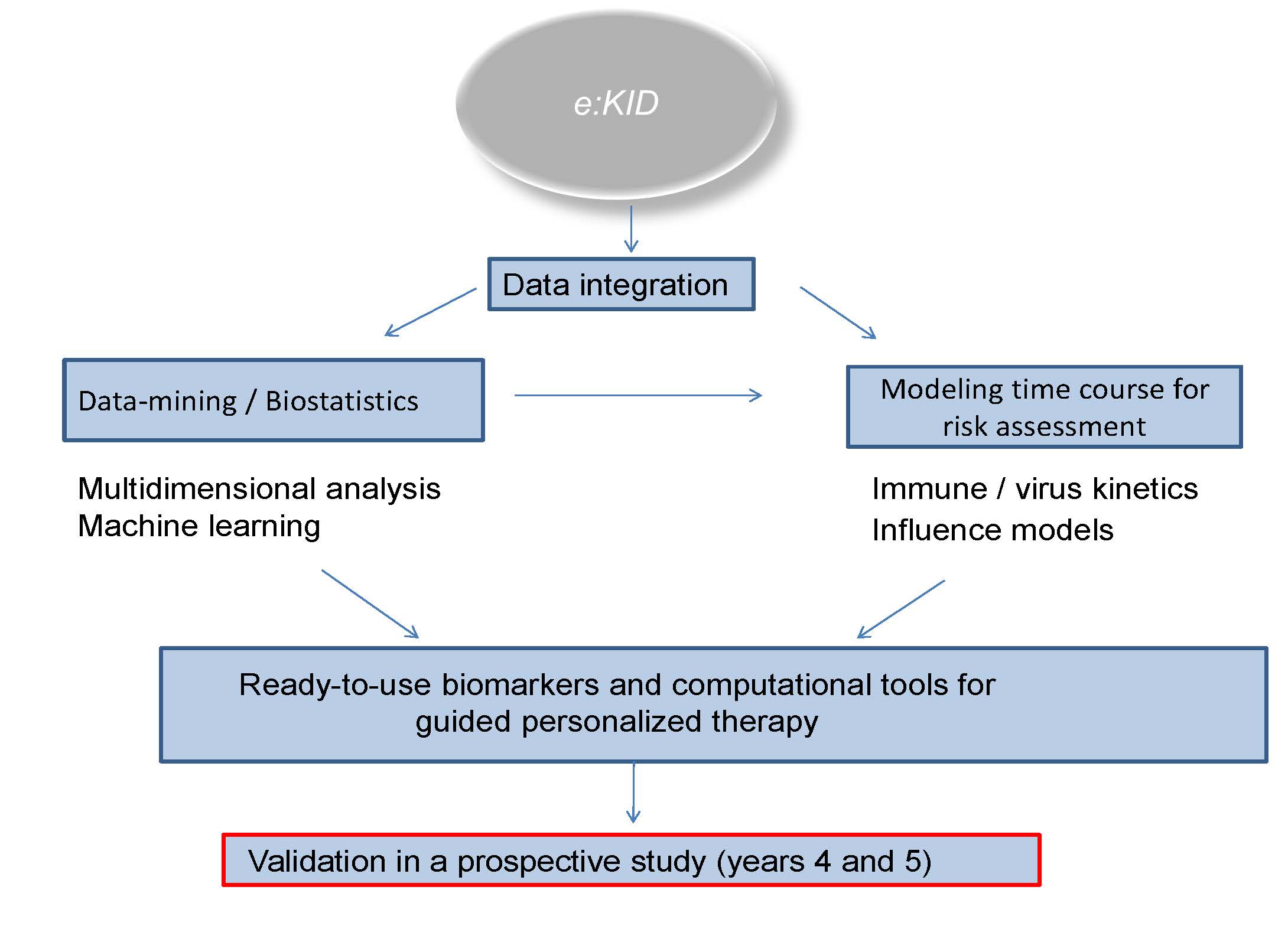
During the first 36 months samples from a completely recruited large multicenter trial incl. a well characterized cohort kidney transplant patients will be analyzed. Based on the proposed mathematical model a prospective clinical trial for personalized adjustment of immunosuppressive therapy will be initiated in the additional 24 months of the project.
Subprojects in e:Kid:
SP 1 Sample management, monitoring of viral infection and virus-specific immunity
SP 3 Assessment of cytokines, the pivotal messengers in intercellular communication
SP 4 Analysis of allo-specific immunity: effector and regulatory T-cells
SP 6 Management of data communication
SP 8 Characterisation of antibody-antigen interactions and adapation on a diagnostic platform
SP 10 Data management and multi-scale data analysis
Keywords: Systems medicine, kidney transplantation, immunosuppressive



