SP 1
Image-guided multiscale modeling of vascular tumor growth as a systems biology-based tool to predict therapeutic outcome in HCC
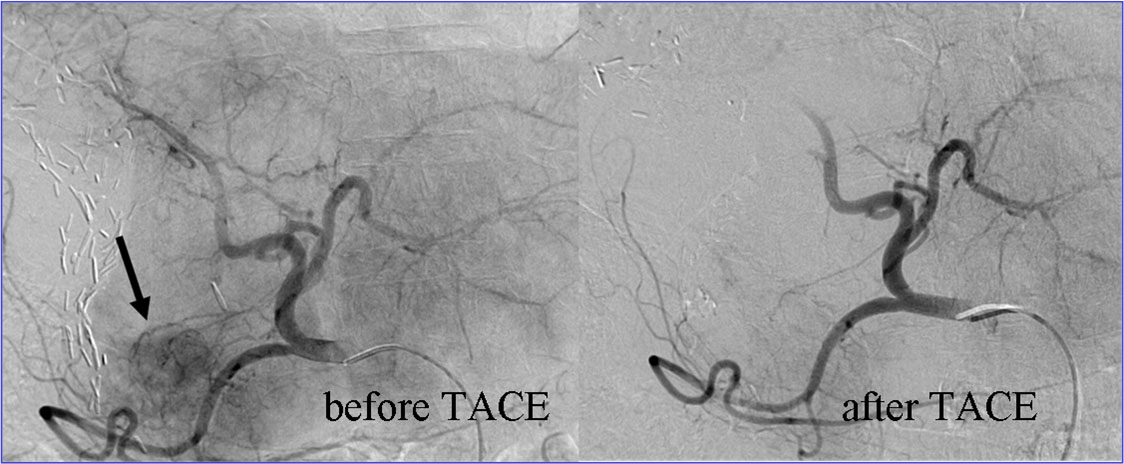
In advanced HCC current established treatment strategies include a modulation of tumor feeding blood vessels. This is accomplished either by drugs that inhibit tumor angiogenesis or by local treatment approaches such as transarterial chemoembolisation (TACE).
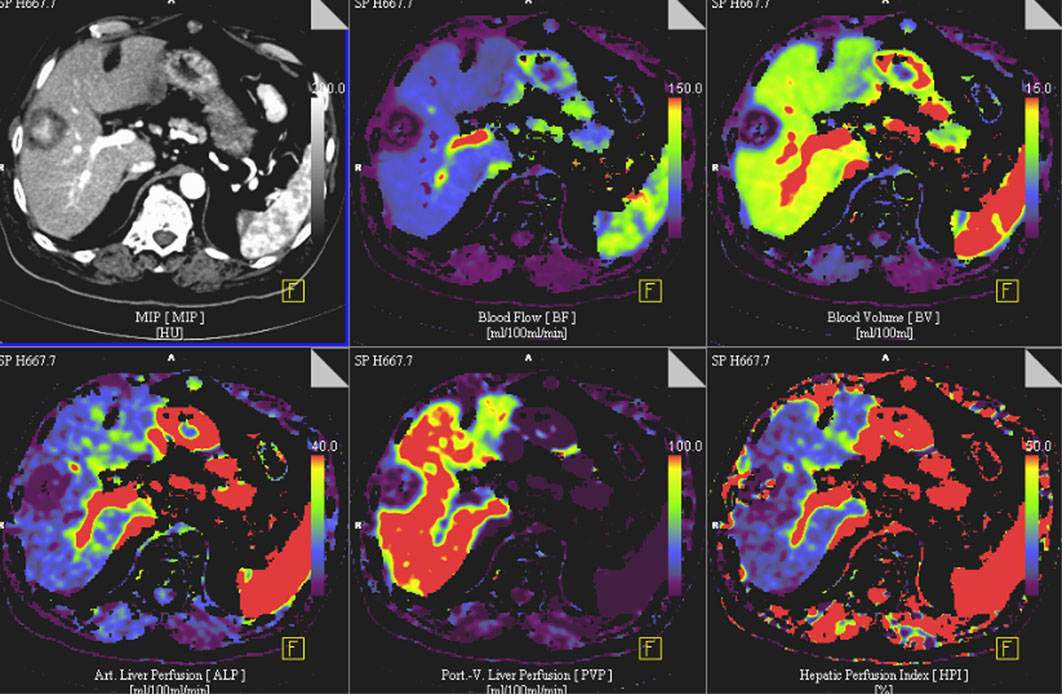

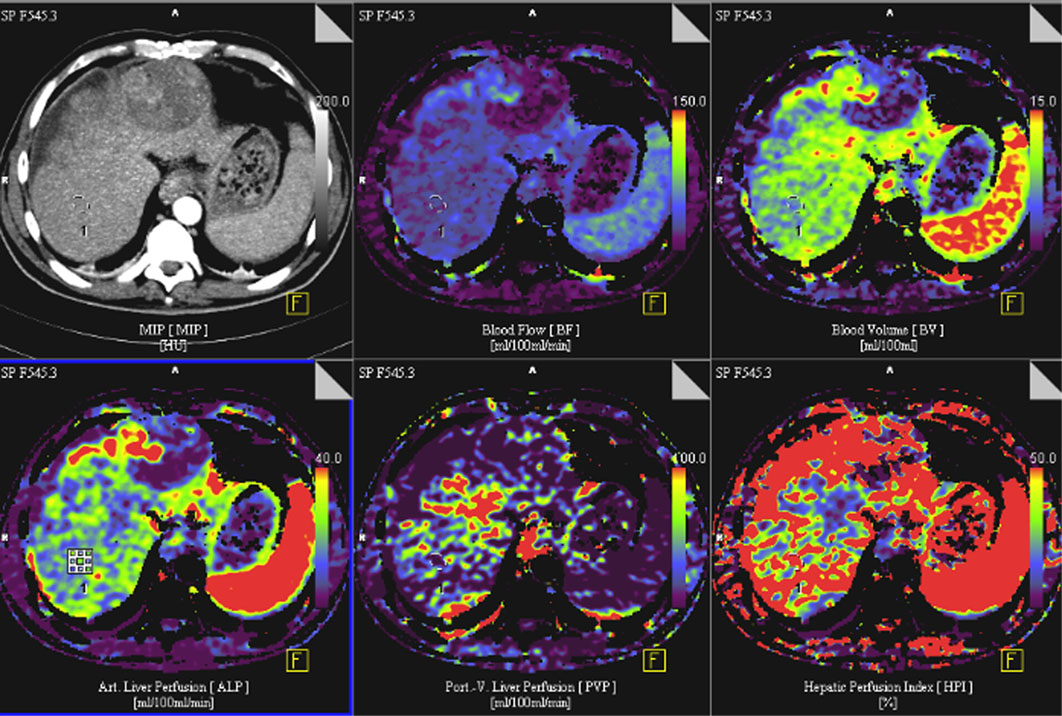
Dynamic modulation of the tumor microcirculation plays a major role with respect to growth characteristics, metastasis and therapy responses. Thus, an in detail characterization of the tumor vascular bed by modern imaging techniques and the integration of this information into a rigorous mathematical model should enable the development of an individualized patient tailored treatment plan, including the supervision of therapy responses. Starting from data sets of already acquired volume perfusion CT (VPCT) and arterial angiography imaging in patients with HCC, a correlation between clinical-radiological data and a 3D multiscale model of vascular tumor growth will be investigated in a first step.

SP4 will provide novel data analysis and mining tools. Subsequently, the imaging fingerprints will be evaluated for their potential to refine and individualize the tumor vasculature prediction model. A connection between this refined model and the identification of relevant biological subnetworks will be performed in SP3. Finally, current clinical open questions for HCC treatments will be simulated, followed by a transfer and validation of the results in a “back-to-the-bedside” approach in SP5.

The ultimate goal of this conjoint effort is to develop a new multiscale model that directly contributes to treatment decisions based on imaging data acquired in daily clinical practice.
Keywords: HCC (hepatozelluläres Karzinom); Volumenperfusions-CT (VPCT); transarterielle Chemoembolisation (TACE)



