SP 8
Chemical Biology of multi-pathway inhibition
In recent years, enormous wealth of knowledge has been accumulated in the identification and understanding of genetic modifications and their negative consequences which lead to the formation of cancer. Despite this, many correlations like the function of involved proteins, the dynamic regulation of cellular behavior and the mechanisms by which cells develop drug resistances are still unknown.
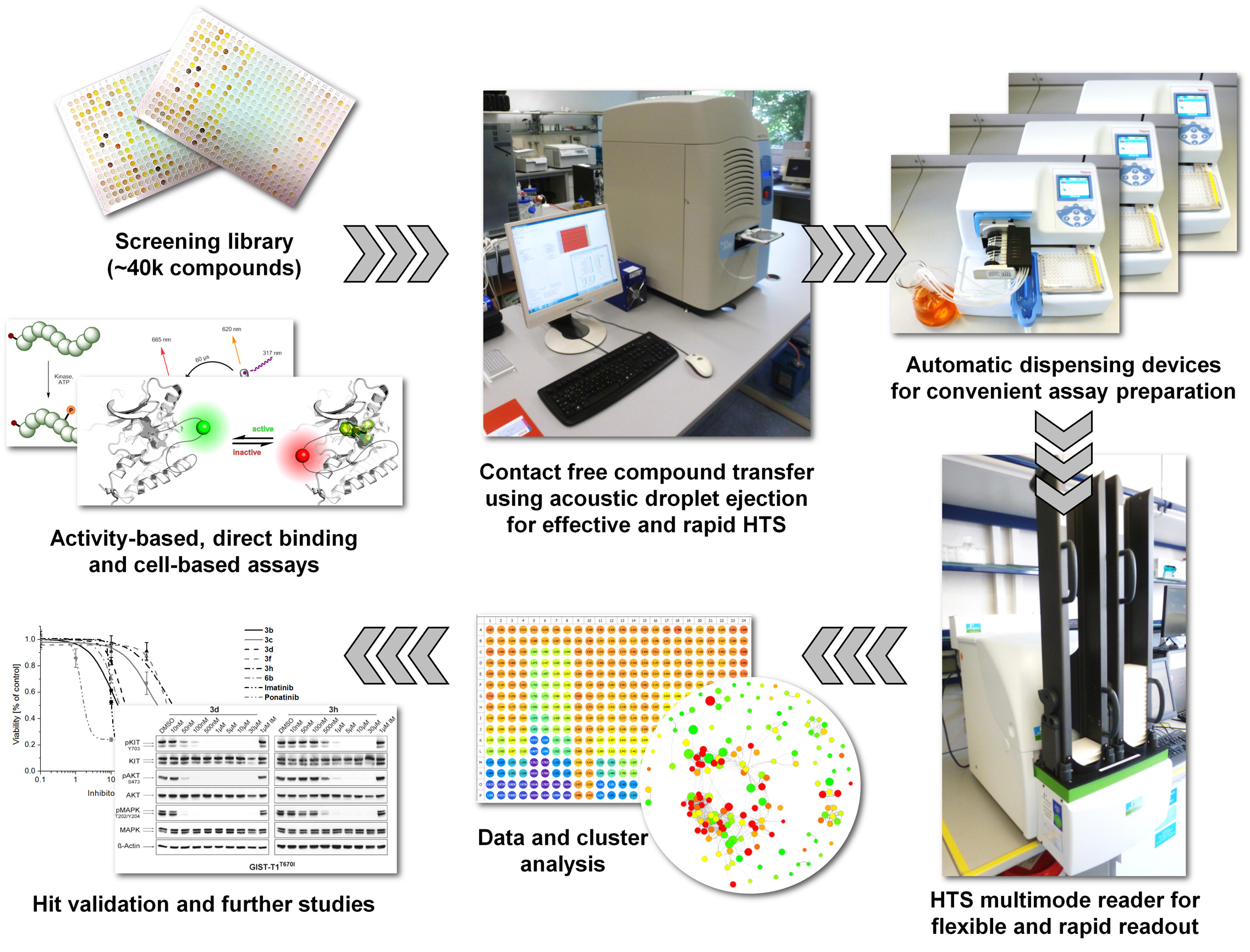
On the base of our knowledge in chemical biology the intention of our project is to identify and characterize proteins, which promote the transformation of healthy cells into cancer cells. To achieve this, we will screen small molecule libraries to identify ligands and/or inhibitors and evaluate their effect on a diverse set of proteins in biochemical studies. These findings will be utilized to optimize the properties of these molecules and to generate suitable tools for subsequent cellular studies. In particular, we will focus on the identification and modulation of key regulators of the cell cycle and programmed cell death, respectively. Protein kinases are an example of such a class of target protein since they are often misregulated in many tumors. Since protein kinases share high structural similarity in their catalytic centers, it is challenging to selectively address a given kinase by small molecule inhibitors. Therefore, we will direct our efforts to targeting binding pockets located in different parts of the proteins which may translate into the selective modulation of kinases.
In a second approach, we aim for the modulation of cancer relevant transcription factors. We will target protein-protein-interactions between transcription factors and regulatory partners. Such interplays maintain the protection of cells against oxidative stress and are abrogated in diverse tumors. Major consequences are enhanced cell division rates and drug resistance, which ultimately accompanies with poor prognosis for the patients. Structure-based design approaches and preparative organic chemistry shall lead to the generation of transcription factor interactor-complex stabilizers. Stabilization of such complexes will increase the rate of ubiquitinylation of the target protein, which in turn should result in enhanced protein degradation and furthermore decreased division rates and drug resistance.